Compare
02/22/2025 2025-02-22 23:41Compare
| Yvoire Contour remove | |||||
|---|---|---|---|---|---|
| Name | Yvoire Contour remove | ||||
| Image | 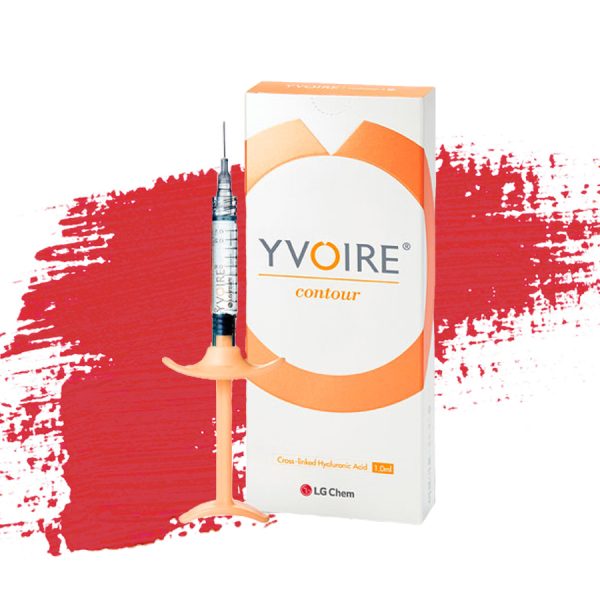 | ||||
| SKU | FM-02-15559 | ||||
| Rating | |||||
| Price | $63.00 | ||||
| Stock | |||||
| Availability | |||||
| Add to cart | |||||
| Description | |||||
| Content | Yvoire Contour – Dermal Fillers Injectable Yvoire Contour is a premium dermal filler designed for deep injection and volumization. It restores structural support, enhances facial definition, and delivers natural, long-lasting contouring results. Key Benefits
Professional Use OnlyYvoire Contour must be administered exclusively by licensed medical professionals or certified aesthetic practitioners. Dosage and injection sites should be customized to each patient’s needs. Additional InformationCountry of Origin: Korea | ||||
| Weight | N/A | ||||
| Dimensions | N/A | ||||
| Additional information |
|